This theory explained the paramagnetic nature of $\text$. 
Tmr thee bond orderrefers to the number of bonds that exist between two atoms.Hint: Molecular orbital theory was put forward by Hund and Mulliken, which can be applied to explain the properties, that was not explained by Valence bond theory. 
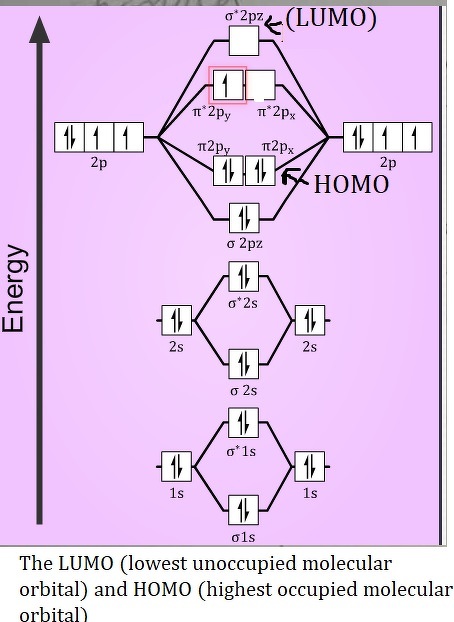
What are the factors that determine orbital interaction?įactors that determine orbital interaction: !energy difference between the interacting orbitals !magnitude of their overlap For the interaction to be strong, th e energies of the two orbitals must b e approximately equal and the overlap must be larg e 2 Bond Order We can also notice the magnetic property of diamagnetic or paramagnetic. Experiments show that each O2 molecule has two unpaired electrons. How many unpaired electrons are there in O2?īy looking at the O2 molecular orbital diagram, we can see that oxygen has BO of 2 because it has 10 bonding and 6 anti-bonding. Check out the following example of O2 MO diagram below! This diagram is a qualitative descriptive tool explaining chemical bonding in molecules in terms of a molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) molecular orbital method in particular. Printable O2 molecular orbital diagrams are available for you to guide your study in the molecular orbital lesson. If we fill each orbital as per Hund’s rule we will see that oxygen is a diradical having two unpaired electrons with the same spin.Īre there any printable O2 molecular orbital diagrams? This can be proven if we look at the molecular orbital diagram of oxygen. Oxygen is paramagnetic mainly because it consists of two unpaired electrons in its last molecular orbital. The valence orbitals in an oxygen atom in a water molecule differ they consist of four equivalent hybrid orbitals that point approximately toward the corners of a tetrahedron (Figure 2). 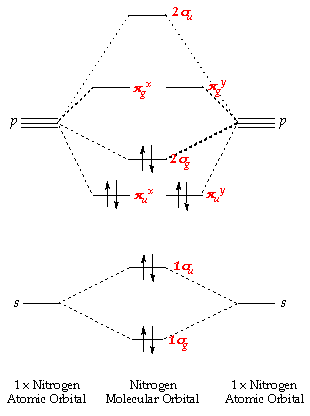
The valence orbitals in an isolated oxygen atom are a 2s orbital and three 2p orbitals. Oxygen/Electron configuration Why is o2 a Diradical? What is the electron configuration for o2? But from the standpoint of theory, dioxygen’s stability is curious: Its highest occupied molecular orbitals contain two unpaired electrons, making it a diradical. Oxygen as O2 is stable enough to be abundant in the environment and is required for many forms of life. Resonance of O2’s π system stabilizes the molecule. In the case of O2- 17 electrons are present &3 electrons are present in antibonding orbitals. Because According to molecular orbital theory O2+ has 15 electrons &it has one electron in antibonding orbital. O2 has two unpaired electrons in its π* orbitals, and a bond order of 2. These 5 atomic orbitals combine to form 10 molecular orbitals. Oxygen molecules will have 16 electrons.Įach oxygen bonds to the other with its 1s, 2s, and 2p orbitals. Thus, two atoms will possess 16 electrons i.e. The molecular orbital theory is a widely accepted theory for describing the electronic structure of molecules. What is the molecular orbital theory for O2? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
